A jump from orbit or step five to orbit three might radiate quanta of red light a jump from six to two might radiate more energy (higher frequency), so it could be a blue light. A jump from step four to step two gives off two steps' worth of energy or a jump from step five to step two gives off three steps of energy.Īn electron jumping to a lower state gives off its energy in the form of light. But Bohr realized, like Planck and Einstein, that this energy can only come in chunks or quanta. In this case, she gives off energy to the floor as she jumps to a lower stair or state. Atoms, too, act in the same way and will not absorb radiation unless the energy they receive contains the minimum to make the next quantum leap.Ī child jumping down stairs also behaves somewhat like an electron changing states within an atom. If she doesn't have quite enough energy to make it to step four, she'll remain at step three.
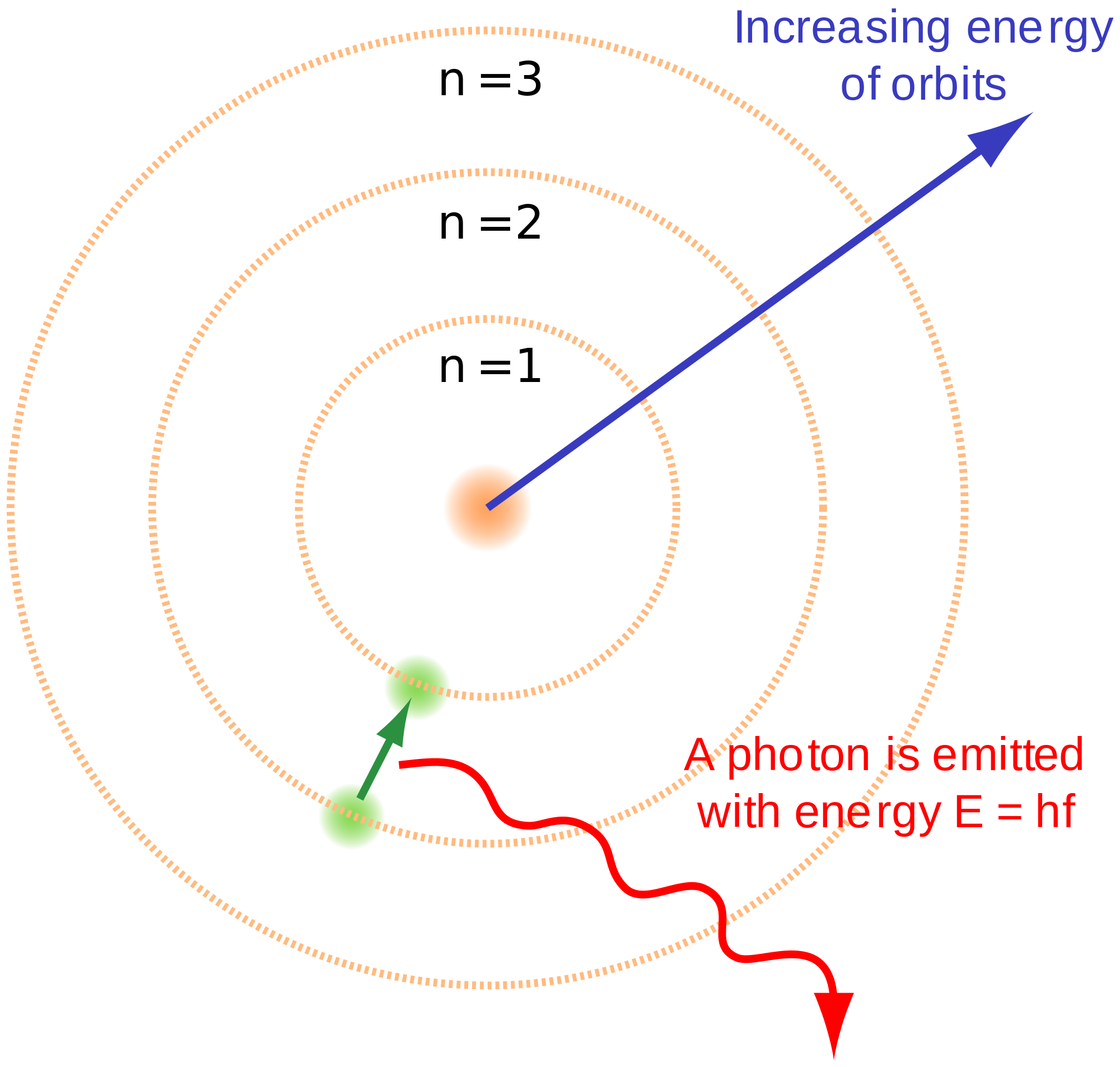
She needs a minimum amount of energy before she can attain the next step or state.

Remember our stair analogy discussed previously? Our child can't land safely or remain stable at step two and one-half, or three and one-third. Cultures, perceptions, beliefs, and even phases of life can often seem discretely separated as individual quantum states, which is why we can feel transformed when we move from one to another. This also seems to embody an irreducible yes/no, on/off quality that lies at the core of computer technology. It seems that some things have to come in whole chunks: children, snowflakes, memories, experiences, paintings, and a whole host of other things. The quantum nature of the universe is not limited to the subatomic world. And if you can imagine an electron acting as a wave (remember that electrons can act as either a particle or a wave) within an atom in much the same way, you can see how it would be forced to assume only a certain number of predetermined vibrational states. It can't vibrate at two and one-half times that frequency. It can vibrate in its fundamental frequency, or twice, three times, or four times that frequency-in other words, its characteristic harmonics. If you pump energy into it and get it swinging, it can vibrate only in a certain number of predetermined ways. You can create your own standing waves by using a jump rope secured at both ends. In the early 1920s, Bohr came up with a way to understand the stability and exactness of atoms using the analogy of standing waves. Remember what Einstein said, "Imagination is more important than knowledge." When you're not really sure how something new operates, its often the best way to begin. However, this is the place where many of the physicists began to formulate their understanding of quantum interactions as well. In most cases, these will suffice to get the ideas involved across even though the theories are much more complex. And when it comes to quantum mechanics, we'll also be explaining these theories and concepts through familiar analogies, metaphors, and other images. We've used our imagination and have done some thought experiments to understand some of the theories we've discussed so far. No one really knew how energy was released, they just knew that it was. As I hope you remember, light is created when energy is released from matter in the form of electromagnetic radiation. The current one that had been developed by Ernst Rutherford needed some tweaking to explain how atoms could emit light and yet not collapse in on themselves. He went about this by developing a better picture of the atom's structure. In order to do that, he would have to explain how energy was released at the atomic level. Having studied the ideas presented by Planck and Einstein, Bohr wanted to theorize about the quantum property of all forms of energy. And some of them have traveled thousands of light years to get to you. The Copenhagen interpretation, which is the standard explanation of what goes on in the quantum worldĭid you know that about 1,000 billion (1012) photons of sunlight fall on a pinhead each second? Even when you look at a faint star, your eye receives a few hundred photons from that star each second.The theory of complementarity, which helps explain wave/particle duality.Bohr's theory of the atom and its quantum structure.

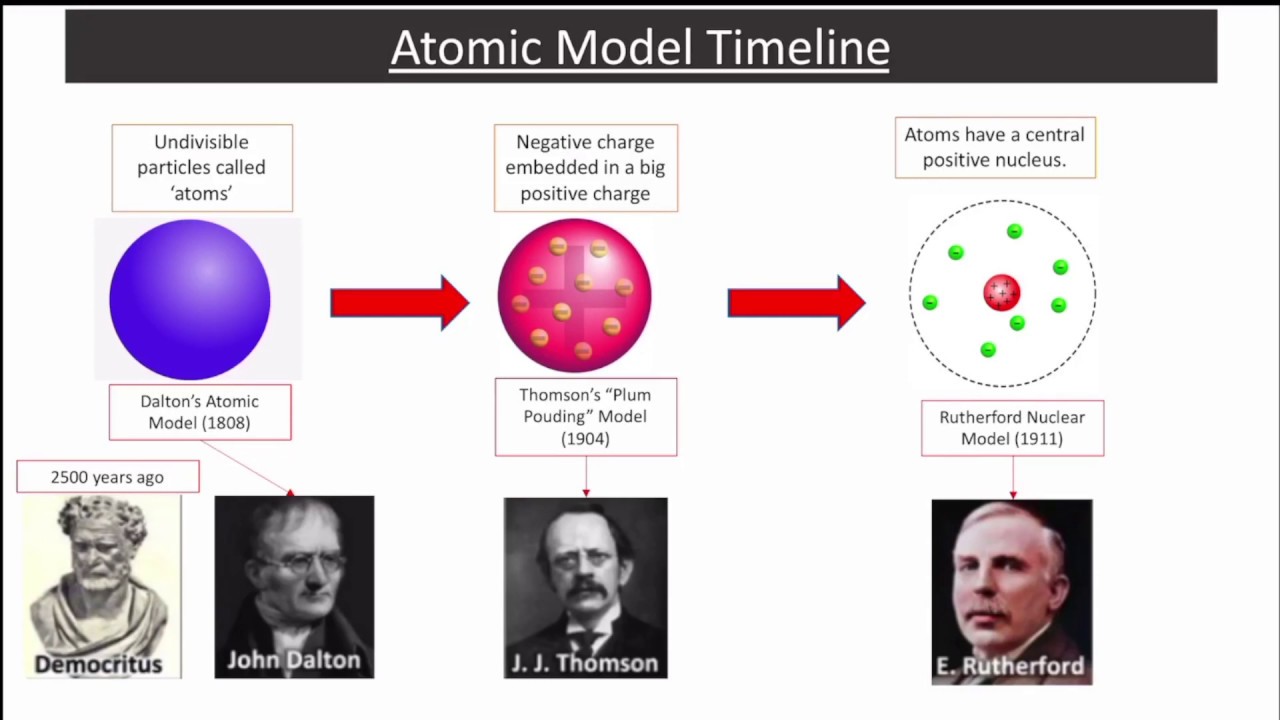
We're now going to cover three of Bohr's most significant contributions (not all in this section, but the next section as well). If Planck is the father of quantum mechanics, then Bohr must be older than him, which in fact he's not, they're not even related-another strange quantum anomaly. Neils Bohr is considered the grandfather of quantum mechanics.

